War Gaming in Drug Development
War gaming, traditionally used in military and business contexts, involves simulating competitive environments to anticipate potential challenges and opportunities. In drug development, war gaming allows teams to proactively identify risks, evaluate different development pathways, and refine strategies to maximize the likelihood of success. By simulating various scenarios, including competitor actions, regulatory hurdles, and market dynamics, war gaming enables informed decision-making and enhances the overall efficiency of the drug development process.
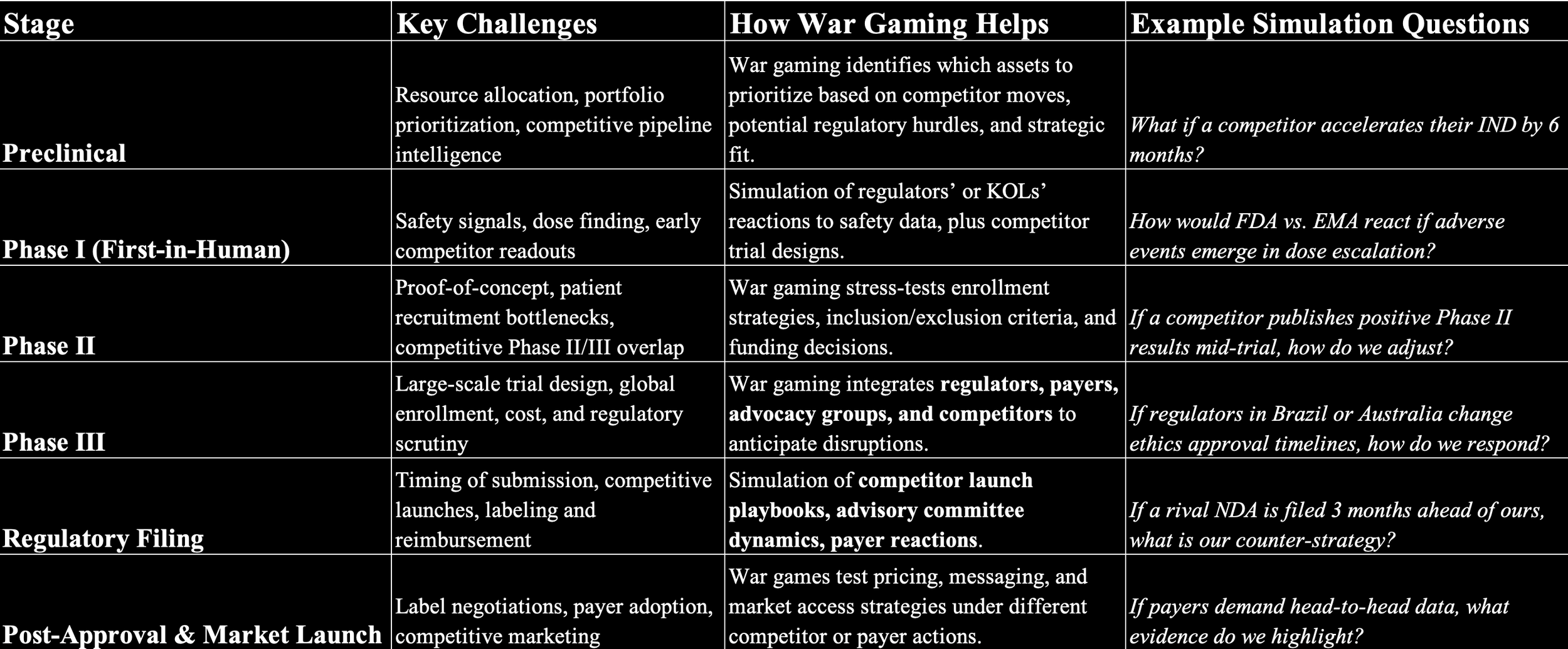
Preclinical
Objective: To optimize target selection, lead identification, and preclinical study design.
War Gaming Scenarios:
Target Validation War Game: Simulate different approaches to validating a drug target, considering potential off-target effects, resistance mechanisms, and alternative therapeutic strategies.
Participants: Scientists, pharmacologists, toxicologists.
Outcomes: Identification of critical experiments, refinement of target validation strategy, and early go/no-go decisions.
Lead Optimization War Game: Explore different chemical modifications and formulations to optimize drug properties (e.g., potency, selectivity, bioavailability).
Participants: Medicinal chemists, formulation scientists, pharmacokineticists.
Outcomes: Prioritization of lead compounds, identification of potential formulation challenges, and optimization of drug delivery strategies.
Preclinical Study Design War Game: Simulate different preclinical study designs to evaluate drug efficacy and safety, considering potential confounding factors and regulatory requirements.
Participants: Preclinical scientists, regulatory affairs specialists, biostatisticians.
Outcomes: Optimization of study protocols, identification of potential safety signals, and improved data interpretation.
Phase 1 Clinical Trials
Objective: To optimize trial design, dose selection, and patient recruitment strategies.
War Gaming Scenarios:
Dose Escalation War Game: Simulate different dose escalation schemes to identify the maximum tolerated dose (MTD) and assess drug safety and tolerability.
Participants: Clinical pharmacologists, clinical investigators, safety physicians.
Outcomes: Optimized dose escalation protocol, identification of potential adverse events, and refined dose selection strategy.
Patient Recruitment War Game: Explore different recruitment strategies to accelerate patient enrollment and ensure trial diversity.
Participants: Clinical operations managers, patient advocacy groups, marketing specialists.
Outcomes: Identification of effective recruitment channels, improved patient engagement, and accelerated trial timelines.
Data Monitoring War Game: Simulate different data monitoring scenarios to identify potential safety signals and ensure data integrity.
Participants: Data managers, biostatisticians, safety physicians.
Outcomes: Enhanced data monitoring plan, early detection of safety signals, and improved data quality.
Phase 2 Clinical Trials
Objective: To optimize trial design, endpoint selection, and patient stratification strategies.
War Gaming Scenarios:
Endpoint Selection War Game: Evaluate different clinical endpoints to assess drug efficacy and demonstrate clinical benefit.
Participants: Clinical investigators, biostatisticians, regulatory affairs specialists.
Outcomes: Identification of relevant and measurable endpoints, improved statistical power, and enhanced clinical trial design.
Patient Stratification War Game: Explore different patient stratification strategies to identify subgroups that are most likely to respond to the drug.
Participants: Clinical investigators, biomarker scientists, geneticists.
Outcomes: Identification of predictive biomarkers, improved patient selection, and enhanced treatment response.
Competitive Landscape War Game: Simulate competitor activities and market dynamics to anticipate potential challenges and opportunities.
Participants: Market research analysts, business development managers, competitive intelligence specialists.
Outcomes: Identification of competitive advantages, refined market positioning, and optimized commercial strategy.
Phase 3 Clinical Trials
Objective: To optimize trial execution, data analysis, and regulatory strategy.
War Gaming Scenarios:
Trial Execution War Game: Simulate potential challenges in trial execution, such as patient retention, data quality issues, and regulatory inspections.
Participants: Clinical operations managers, quality assurance specialists, regulatory affairs specialists.
Outcomes: Proactive mitigation of potential risks, improved trial execution, and enhanced data integrity.
Data Analysis War Game: Explore different statistical analysis plans to maximize the likelihood of demonstrating statistically significant and clinically meaningful results.
Participants: Biostatisticians, clinical investigators, regulatory affairs specialists.
Outcomes: Optimized statistical analysis plan, enhanced data interpretation, and improved regulatory submission.
Regulatory Strategy War Game: Simulate different regulatory pathways and potential interactions with regulatory agencies to optimize the regulatory submission strategy.
Participants: Regulatory affairs specialists, medical writers, legal counsel.
Outcomes: Optimized regulatory pathway, proactive management of regulatory risks, and accelerated approval timelines.
NDA Filing and Post-Market
Objective: To optimize the NDA submission, address potential regulatory queries, and develop a robust post-market strategy.
War Gaming Scenarios:
NDA Submission War Game: Simulate the NDA review process and anticipate potential questions from regulatory agencies.
Participants: Regulatory affairs specialists, medical writers, clinical investigators.
Outcomes: Proactive preparation for regulatory queries, improved NDA submission quality, and accelerated approval timelines.
Market Access War Game: Explore different pricing and reimbursement strategies to optimize market access and ensure commercial success.
Participants: Market access specialists, health economists, pricing analysts.
Outcomes: Optimized pricing strategy, improved market access, and enhanced commercial viability.
Post-Market Surveillance War Game: Simulate potential post-market safety signals and develop a proactive risk management plan.
Participants: Safety physicians, pharmacovigilance specialists, regulatory affairs specialists.
Outcomes: Proactive risk management, enhanced patient safety, and improved product lifecycle management.